This article will be updated regularly through November.
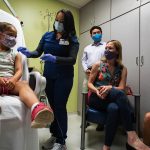
Children 5 to 11 years old can now be vaccinated against COVID-19, after the Food and Drug Administration granted emergency authorization to use a low-dose Pfizer-BioNTech vaccine.
Vaccinations became available starting November 3, as many parents are eager to get their kids vaccinated as the holidays approach. The Biden administration has begun shipping doses for 28 million children.
“We urge parents to get ready and make a plan, and the program will be fully up and running the week of November 8,” Jeffrey Zients, the White House coronavirus response coordinator, told NPR. Zients told NPR that the White House would initially distribute 15 million vaccines.
Pfizer-BioNTech’s smaller vaccine dose for this age group is 90.7% effective at preventing symptomatic COVID-19 infections, the companies said in trial data submitted to the FDA. Pfizer is the first of the three FDA-approved COVID-19 vaccine manufacturers to share trial results for kids ages 5 to 11. The FDA panel’s decision means from the agency’s perspective, the benefits of the vaccine outweigh the risks.
Kids in the trial also contracted the coronavirus as the highly transmissible Delta variant was spreading through the country from July to September, Pfizer and BioNTech said. No cases of severe illness were reported in the trial. The Pfizer-BioNTech vaccine already had emergency use authorization for children ages 12 to 15.
The responsibility of deciding how and whether children obtain COVID-19 vaccinations may largely fall to mothers: They usually make the health decisions about children, per Kaiser Family Foundation (KFF) polling from this spring.
Furthermore, outbreaks in schools have led to classroom quarantines and, in some cases, a return to remote learning, further disrupting life not just for students but also for parents — often mothers, who have shouldered so much of the child care throughout the pandemic.
The 19th spoke with public health experts (who are also moms) about what parents should know as Pfizer and BioNTech seek emergency FDA authorization for young children.
What happens next?
Find available vaccine appointments in your area here, and select Pfizer-BioNTech for kids 5 through 11, as this manufacturer is the only one so far to be reviewed by the FDA and CDC.
The White House is focused on distributing vaccines to pediatric and primary care providers, as well as children’s hospitals and local pharmacies, where families already seek care for their children, Zients told NPR.
The White House said in October that it has acquired enough vaccine to supply more than 25,000 pediatric and primary care providers, and plans to work to set up vaccination sites through the end of 2021.
Parents of children who are younger — those from 2 to 5 years old and infants as young as 6 months old — will have to wait longer. Trial data on these groups is not expected to be released until later this year, Pfizer said in September.
For parents wanting more advice on getting their kids vaccinated, “This is a great time for people to start talking to their pediatrician,” said Margaret Aldrich, director of pediatric infection control at the Children’s Hospital at Montefiore in the Bronx. Some cities like New York and Washington have reopened mass vaccination sites, and vaccine appointments are also available at some schools.
Natasha Burgert, a general pediatrician in Overland Park, Kansas, told The 19th she is encouraging vaccine-hesitant parents to talk to their pediatricians and other trusted sources to make decisions for their family, especially as misinformation spreads online.
“That’s LOTS of critical eyes on the data set to make sure the science is sound,” she wrote in a newsletter to parents in September. “There’s nobody out there that’s trying to make or push this vaccine, especially onto children, without due diligence,” she told The 19th.
Do kids of different ages get different doses?
Yes. The Pfizer-BioNTech COVID-19 vaccine being given to kids over 12 uses a 30-microgram dose, while doses of only 10 micrograms, administered 21 days apart, are effective (and create a robust antibody response) for children 5 to 11, the companies said.
Aren’t children less likely to get COVID-19?
The peak of child COVID-19 cases in the U.S. was seen in early September, per the American Academy of Pediatrics, and has steadily decreased since then, although the overall count of newly diagnosed children remains “extremely high,” the organization says in its latest data report.
Although children experiencing severe illness from COVID-19 is uncommon, and many may not display symptoms, the need to protect children is still high. Hospital pediatric units met capacity this fall across the country and in cities like Pittsburgh and Fort Worth, Texas, as the new school year kicked off. Pediatric hospitalizations caused by the virus also increased sharply this summer as the Delta variant spread.
“Once you have a large number of kids … getting COVID, it’s a numbers game. Some of those kids are going to run into trouble. Kids have needed ICU-level care,” Aldrich told The 19th. “Kids have had complications related to COVID over the past many months now.”
A KFF survey in August found that 4 in 10 parents of children under 12 wanted to “wait and see” before getting their child vaccinated against the virus, after a COVID-19 vaccine is authorized and available for their kids. A majority of those parents said they were most concerned about their children experiencing serious side effects from the vaccine and that there is not enough information about long-term effects.
“The risk of death for COVID far outweighs the risks associated with this vaccine, as was shown for the adult clinical trials,” said Aldrich, who is a mother of three daughters. Her oldest is currently vaccinated, and she plans to have her other children inoculated as soon as it’s available for them.
Burgert said she’s looking forward to when her unvaccinated 11-year-old son can be protected against the virus, as her 14-year-old daughter is already vaccinated.
A statistically insignificant number of cases of myocarditis, an inflammatory heart condition, have been seen so far in clinical trials of the COVID vaccine in kids, The 19th reported last month. In most cases, this very rare side effect began within a few days after receiving a second Pfizer dose. This follows a greater history of delayed vaccine side effects, which tend to happen within two months of vaccination.
“The process that this vaccine has gone through really has been consistent with the process that any vaccine would go through,” Aldrich said.
“This is going to seem fast, but it’s really not,” Burgert wrote in her Monday newsletter. “We are in a global pandemic. As such, we have the luxury of nearly unlimited cash AND the full attention of an international collection of brilliant minds.”
Although the risk for children getting seriously ill from the virus is lower than for older Americans, Aldrich stressed that “if you are the one person who’s child ends up in the ICU, that’s 100 percent of your experience. If you can prevent that from happening, why wouldn’t you do that?”
I’m a working parent. How can I make time to get my kid vaccinated?
The Pfizer vaccine still requires two shots — and if children experience mild side effects like chills or fever, they could have to miss school and need care at home. Employed parents may have to take time off from work to care for their children — a responsibility that 61 percent of employed mothers told KFF this spring usually falls to them.
Few working parents told KFF in August that their employer would offer paid time off to get their children vaccinated, or paid time off to care for them if they experience short-term side effects.
If time off isn’t possible, there are other options — like relying on a family member or trusted guardian to go with your child to the vaccination appointment.
“Most pediatricians and public health places will accept a nanny or a babysitter or a grandparent to bring them to the clinic with an authorized letter that says that they are authorized to be able to sign on behalf of that patient,” Burgert said.
Will my school mandate COVID-19 vaccines?
It depends on where you live. In California, Gov. Gavin Newsom has said that eligible California students must get vaccinated against the coronavirus in order to take classes in-person, pending full FDA approval of shots for students’ age groups. The governor’s office reportedly said that they don’t expect the mandate to take affect until at least the next school year.
Zients, the White House coronavirus response coordinator, also stressed on October 20 that vaccine mandates for children depend on local school district and state decisions, although the Biden administration supports such vaccine requirements.
“School vaccination requirements have been around for decades, and those decisions should be made at the state and local level,” Zients said. “We know that in general, requirements work, and we support states and school districts taking actions to ensure that everyone who’s eligible get vaccinated.”
How can I protect my child now?
For families gathering for Thanksgiving, CDC director Rochelle Walensky said in early November that adults and all eligible people in the house should be vaccinated — and for young children that are newly eligible,
“As more and more people around the table are vaccinated, it will be safer not just for those who are vaccinated, for those who are unable to be vaccinated, our children under 5, or potentially those who are immunocompromised,” she said.